Iodine-131
| Iodine-131 | |
|---|---|
| General | |
| Name, symbol | Radioiodine,131I |
| Neutrons | 78 |
| Protons | 53 |
| Nuclide data | |
| Half-life | 8.0197 days |
| Isotope mass | 130.9061246(12) u |
| Excess energy | mc2
1 ss keV |
Iodine-131 (131I), also called radioiodine (though many other radioactive isotopes of this element are known), is an important radioisotope of iodine. It has a radioactive decay half-life of about eight days. Its uses are mostly medical and pharmaceutical. It also plays a major role as a radioactive isotope present in nuclear fission products, and was a significant contributor to the health hazards from open-air atomic bomb testing in the 1950s, and from the Chernobyl disaster, as well as being a large fraction of the contamination hazard in the first weeks in the Japanese nuclear crisis. This is because I-131 is a major uranium, plutonium fission product, comprising nearly 3% of the total products of fission (by weight). See fission product yield for a comparison with other radioactive fission products. I-131 is also a major fission product of uranium-233, produced from thorium.
Due to its mode of beta decay, iodine-131 is notable for causing mutation and death in cells that it penetrates, and other cells up to several millimeters away. For this reason, high doses of the isotope are sometimes less dangerous than low doses, since they tend to kill thyroid tissues that would otherwise become cancerous as a result of the radiation. For example, children treated with moderate dose of I-131 for thyroid adenomas had a detectable increase in thyroid cancer, but children treated with a much higher dose did not. Likewise, most studies of very-high-dose I-131 for treatment of Graves disease have failed to find any increase in thyroid cancer, even though there is linear increase in thyroid cancer risk with I-131 absorption at moderate doses.[1] Thus, iodine-131 is increasingly less employed in small doses in medical use (especially in children), but increasingly is used only in large and maximal treatment doses, as a way of killing targeted tissues. This is known as "therapeutic use."
Iodine-131 can be "seen" by nuclear medicine imaging techniques (i.e., gamma cameras) whenever it is given for therapeutic use, since about 10% of its energy and radiation dose is via gamma radiation. However, since the other 90% of radiation (beta radiation) causes tissue damage without contributing to any ability to see or "image" the isotope, other less-damaging radioisotopes of iodine are preferred in situations when only nuclear imaging is required. The isotope I-131 is still occasionally used for purely diagnostic (i.e., imaging) work, due to its low expense compared to other iodine radioisotopes. Very small medical imaging doses of I-131 have not shown any increase in thyroid cancer. The low-cost availability of I-131, in turn, is due to the relative ease of creating I-131 by neutron bombardment of natural tellurium in a nuclear reactor, then separating I-131 out by various simple methods (i.e., heating to drive off the volatile iodine). By contrast, other iodine radioisotopes are usually created by far more expensive techniques, starting with reactor radiation of expensive capsules of pressurized xenon gas.
Much smaller incidental doses of iodine-131 than are used in medical therapeutic uses, are thought to be the major cause of increased thyroid cancers after accidental nuclear contamination.[2] These cancers happen from residual tissue radiation damage caused by the I-131, and usually appear years after exposure, long after the I-131 has decayed.
Contents |
Production
Most I-131 production is from nuclear reactor neutron-irradiation of a natural tellurium target. Irradiation of natural tellurium produces almost entirely I-131 as the only radionuclide with a half-life longer than hours, since most lighter isotopes of tellurium become heavier stable isotopes, or else stable iodine or xenon. However, the heaviest naturally-occurring tellurium nuclide, Te-130 (34% of natural Te) absorbs a neutron to become tellurium-131, which beta-decays with a half-life of 25 minutes, to I-131.
A tellurium compound can be irradiated while bound as an oxide to an ion exchange column, and evolved I-131 then eluted into an alkaline solution.[3] More commonly, powdered elemental tellurium is irradiated and then I-131 separated from it by dry distillation of the iodine, which has a far higher vapor pressure. The element is then dissolved in a mildly alkaline solution in the standard manner, to produce I-131 as iodide and hypoiodate (which is soon reduced to iodide).[4]
Radioactive decay
131I decays with a half-life of 8.02 days with beta and gamma emissions. This nuclide of iodine atom has 78 neutrons in nucleus, the stable nuclide 127I has 74 neutrons. On decaying, 131I most often (89% of the time) expends its 971 keV of decay energy by transforming into the stable 131Xe (Xenon) in two steps, with gamma decay following rapidly after beta decay:
 + 606 keV
+ 606 keV
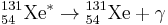 + 364 keV
+ 364 keV
The primary emissions of 131I decay are thus beta particles with a maximal energy of 606 keV (89% abundance, others 248 – 807 keV) and 364 keV gamma rays (81% abundance, others 723 keV).[5] Beta decay, as always in this process, also produces an antineutrino, which carries off variable amounts of the beta decay energy.
The beta particles, due to their high mean energy (190 keV, with typical beta-decay spectra present) have a tissue penetration of 0.6 to 2 mm.[6]
131I is a fission product with a yield of 2.878% from uranium-235,[7] and can be released in nuclear weapons tests and nuclear accidents. However, the short half-life means it is not present in significant quantities in cooled spent nuclear fuel, unlike iodine-129 whose half-life is nearly a billion times that of I-131.
Effects of exposure
Iodine in food is absorbed by the body and preferentially concentrated in the thyroid where it is needed for the functioning of that gland. When 131I is present in high levels in the environment from radioactive fallout, it can be absorbed through contaminated food, and will also accumulate in the thyroid. As it decays, it may cause damage to the thyroid. The primary risk from exposure to high levels of 131I is the chance occurrence of radiogenic thyroid cancer in later life. Other risks include the possibility of non-cancerous growths and thyroiditis.[1]
The risk of thyroid cancer in later life appears to diminish with increasing age at time of exposure. Most risk estimates are based on studies in which radiation exposures occurred in children or teenagers. When adults are exposed, it has been difficult for epidemiologists to detect a statistically significant difference in the rates of thyroid disease above that of a similar but otherwise-unexposed group.[1]
The risk can be mitigated by taking iodine supplements, raising the total amount of iodine in the body and, therefore, reducing uptake and retention in tissues and lowering the relative proportion of radioactive iodine. However, such supplements were not distributed to the population living nearest to the Chernobyl nuclear power plant after the disaster,[8] though they were widely distributed to children in Poland.
Within the USA, the highest 131I fallout doses occurred during the 1950s and early 1960s to children having consumed fresh sources of milk contaminated as the result of above-ground testing of nuclear weapons.[9] The National Cancer Institute provides additional information on the health effects from exposure to 131I in fallout,[10] as well as individualized estimates, for those born before 1971, for each of the 3070 counties in the USA. The calculations are taken from data collected regarding fallout from the nuclear weapons tests conducted at the Nevada Test Site.[11]
On 27 March 2011, the Massachusetts Department of Public Health reported that 131I was detected in very low concentrations in rainwater from samples collected in Massachusetts, USA, and that this likely originated from the Fukushima power plant.[12] Farmers near the plant dumped raw milk, while testing in the United States found 0.8 pico-curies per liter of iodine-131 in a milk sample, but the radiation levels were 5,000 times lower than the FDA's "defined intervention level." The levels were expected to drop relatively quickly [13]
Treatment and prevention
A common treatment method for preventing iodine-131 exposure is by saturating the thyroid with regular, non-radioactive iodine-127, as an iodide salt. Free elemental iodine should not be used for saturating the thyroid because it is a corrosive oxidant and therefore is toxic to ingest in the necessary quantities. The thyroid will absorb very little of the radioactive iodine-131 after it is saturated with non-radioactive iodide, thereby avoiding the damage caused by radiation from radioiodine. The most common method of treatment is to give potassium iodide to those at risk. The dosage for adults is 130 mg potassium iodide per day, given in one dose, or divided into portions of 65 mg twice a day. This is equivalent to 100 mg of iodide, and is about 700 times bigger than the nutritional dose of iodide, which is 0.15 mg per day (150 micrograms per day). See potassium iodide for more information on prevention of radioiodine absorption by the thyroid during nuclear accident, or for nuclear medical reasons.
The ingestion of prophylaxis iodide & iodate is not without its dangers, There is reason for caution about taking potassium iodide or iodine supplements, as their unnecessary use can cause conditions such as the Jod-Basedow phenomena, and the Wolff-Chaikoff effect, trigger and/or worsen hyperthyroidism and hypothyroidism, and ultimately cause temporary or even permanent thyroid conditions. It can also cause sialadenitis (an inflammation of the salivary gland), gastrointestinal disturbances, allergic reactions and rashes. Potassium iodide is also not recommended for those who have had an allergic reaction to iodine, and people with dermatitis herpetiformis and hypocomplementemic vasculitis, conditions that are linked to a risk of iodine sensitivity. [14]
The administration of known goitrogen substances can also be used as a prophylaxis in reducing the bio-uptake of iodine (whether it be non-radioactive iodine-127 or radioactive iodine-131, as the body cannot discern between the different iodine isotopes). Perchlorate ions, a common water contaminant in the USA due to the aerospace industry, has been shown to reduce iodide uptake. Perchlorate is a competitive inhibitor of the process by which iodide, is actively deposited into thyroid follicular cells. A study involving healthy adult volunteers determined that at levels above 0.007 milligrams per kilogram per day (mg/(kg·d)), perchlorate begins to temporarily inhibit the thyroid gland’s ability to absorb iodine from the bloodstream ("iodide uptake inhibition", thus perchlorate is a known goitrogen). The purposeful addition of ~ 250 ppb of perchlorate ions to a regions water supply, for approximately three months, immediately after a radioiodine release, could thus be beneficial to the population in preventing radioiodine bioaccumulation, independent of the availability of Iodate or Iodide drugs[15]. In the event of a radioiodine release the ingestion of potassium iodide or iodate, if available, would rightly take precedence and would be the first line of defense in protecting the population from a radioiodine release. However in the event of a radioiodine release too massive and widespread to be mediation by the limited stock of iodide & iodate prophylaxis drugs, then the addition of perchlorate ions to the water supply would serve as a cheap, efficacious, second line of defense against radioiodine bioaccumulation.
The ingestion of goitrogen drugs is also not without its dangers, such as hypothyroidism. In all these cases however, despite the risks, the prophylaxis benefits of intervention with iodide, iodate and perchlorate outweigh the serious cancer risk from radioiodine bioaccumulation in conditions of radioiodine contamination of the environment.
Medical and pharmaceutical uses
It is used in nuclear medicine therapeutically and can also be seen with diagnostic scanners if it has been used therapeutically. Use of the 131I as iodide salt exploits the mechanism of absorption of iodine by the normal cells of the thyroid gland. Examples of its use in radiation therapy are those where tissue destruction is desired after iodine uptake by the tissue.
Major uses of 131I include the treatment of thyrotoxicosis (hyperthyroidism) and some types of thyroid cancer that absorb iodine. The 131I is thus used as direct radioisotope therapy to treat hyperthyroidism due to Graves' disease, and sometimes hyperactive thyroid nodules (abnormally active thyroid tissue that is not malignant). The therapeutic use of radioiodine to treat hyperthyroidism from Graves' disease was first reported by Saul Hertz in 1941.
The 131I isotope is also used as a radioactive label for certain radiopharmaceuticals that can be used for therapy, e.g. 131I-metaiodobenzylguanidine (131I-MIBG) for imaging and treating pheochromocytoma and neuroblastoma. In all of these therapeutic uses, 131I destroys tissue by short-range beta radiation. About 90% of its radiation damage to tissue is via beta radiation, and the rest occurs via its gamma radiation (at a longer distance from the radioisotope). It can be seen in diagnostic scans after its use as therapy, because 131I is also a gamma-emitter.
Because of the carcinogenicity of its beta radiation in the thyroid in small doses, I-131 is rarely used primarily or solely for diagnosis (although in the past this was more common due to this isotope's relative ease of production and low expense). Instead the more purely gamma-emitting radioiodine Iodine-123 is used in diagnostic testing (nuclear medicine scan of the thyroid). The longer half-lived iodine-125 is also occasionally used when a longer half-life radioiodine is needed for diagnosis, and, in brachytherapy treatment (isotope confined in small seed-like metal capsules), where the low-energy gamma radiation without a beta component, makes iodine-125 useful. The other radioisotopes of iodine are never used in brachytherapy.
The use of 131I as a medical isotope has been blamed for a routine shipment of biosolids being rejected from crossing the Canada—U.S. border.[16] Such material can enter the sewers directly from the medical facilities, or by being excreted by patients after a treatment.
Post-treatment isolation
Patients receiving I-131 radioiodine treatment are warned not to have sexual intercourse for one month (or shorter, depending on dose given), and women are told not to become pregnant for six months afterwards. "This is because a theoretical risk to a developing fetus exists, even though the amount of radioactivity retained may be small and there is no medical proof of an actual risk from radioiodine treatment. Such a precaution would essentially eliminate direct fetal exposure to radioactivity and markedly reduce the possibility of conception with sperm that might theoretically have been damaged by exposure to radioiodine."[17] These guidelines vary from hospital to hospital and will depend also on the dose of radiation given. Some also advise not to hug or hold children when the radiation is still high, and a one or two metre distance to others may be recommended.[18]
I-131 will be eliminated from the body over the next several weeks after it is given. The majority of I-131 will be eliminated from the human body in 3–5 days, through natural decay, and through excretion in sweat and urine. Smaller amounts will continue to be released over the next several weeks, as the body processes thyroid hormones created with the I-131. For this reason, it is be advised to regularly clean toilets, sinks, bed sheets and clothing used by the person who received the treatment. Patients may also be advised to wear slippers or socks at all times, and themselves physically isolated from others. This minimizes accidental exposure by family members, especially children.[19] Use of a decontaminant specially made for radioactive iodine removal may be advised. The use of chlorine bleach solutions, or cleaners that contain chlorine bleach for cleanup, are not advised, since radioactive elemental iodine gas may be released.[20] Airborne I-131 may cause a greater risk of second-hand exposure, spreading contamination over a wide area.
Many airports now have radiation detectors to detect the smuggling of radioactive materials that may be used in nuclear weapons manufacture. Patients should be warned that if they travel by air, they may trigger radiation detectors at airports up to 95 days after their treatment with 131I.[21]
See also
References
- ^ a b c The Management of Graves’ Disease in Children, with Special Emphasis on Radioiodine Treatment. Scott A. Rivkees, Charles Sklar and Michael Freemark J. Clin. Endocrinol. Metab. 1998 83: 3767-3776, doi: 10.1210/jc.83.11.3767. For full text see: [1]. Quote:
The increased risk of thyroid cancer after thyroid irradiation in childhood has been recognized for nearly 50 yr (119). Thus, a major concern of iodine-131 therapy relates to the risks of thyroid and nonthyroid cancers. However, this issue has been the focus of several long term follow-up studies involving more than 60,000 patients (56, 120–123). Studies of the effects of external radiation, diagnostic iodine- 131 use, and environmental radioiodine and g-ray exposure have also provided important insights regarding the risks of radiation exposure and thyroid carcinomas (26, 28, 124–128). These studies show that the risk of thyroid cancer is increased with exposure to low or moderate levels of external radiation. In contrast, thyroid cancer risks are much lower after high-level irradiation that results in thyroid cell death or reduced capacity of cells to divide (128, 129).
[...]
The large-scale epidemiological surveys of the CTSG involving 36,050 patients in the United States (56) and the Swedish cohort studies (16, 121, 141) have provided considerable information about the relative cancer risks after iodine-131 therapy. After treatment of Graves’ disease in adults with iodine-131, which exposes the thyroid gland to high levels of radiation, rates of thyroid cancer and thyroid cancer mortality were not increased (56, 121, 141, 160).
Follow-up data involving children in the CTSG showed that thyroid adenomas developed in 30% of the patients treated in one center with low doses of iodine-131 (50 mCi/g) estimated to result in thyroid exposure of 2500 cGy (56, 88). Yet, in the other centers where children were treated with higher doses of iodine-131 (100–200 mCi/g), the incidence of thyroid neoplasms was not increased (56).
- ^ [2] National Cancer Institute calculator for thyroid cancer risk as a result of I-131 intake after nuclear testing before 1971 in Nevada
- ^ Chattopadhyay, Sankha; Saha Das, Sujata (2010). "Recovery of 131I from alkaline solution of n-irradiated tellurium target using a tiny Dowex-1 column". Applied Radiation and Isotopes 68 (10): 1967–9. doi:10.1016/j.apradiso.2010.04.033. PMID 20471848.
- ^ "I-131 Fact Sheet". http://www.mds.nordion.com/documents/products/I-131_Solu_Can.pdf. Retrieved 2010-10-26. see this page for modern production technique
- ^ "Nuclide Safety Data Sheet". http://hpschapters.org/northcarolina/NSDS/131IPDF.pdf. Retrieved 2010-10-26.
- ^ Mario Skugor (2006). Thyroid Disorders (A Cleveland Clinic Guide). Cleveland Clinic Press. pp. 82. ISBN 9781596240216.
- ^ "Nuclear Data for Safeguards, Table C-3, Cumulative Fission Yields". International Atomic Energy Agency. http://www-nds.iaea.org/sgnucdat/c3.htm. Retrieved 14 March 2011. (thermal neutron fission)
- ^ http://www.ecolo.org/documents/documents_in_english/Causes.ChernobyJF.doc
- ^ Simon, Steven; Bouville, André; Land, Charles (2006). "Fallout from Nuclear Weapons Tests and Cancer Risks". American Scientist 94: 48. doi:10.1511/2006.1.48.
- ^ "Radioactive I-131 from Fallout". National Cancer Institute. http://www.cancer.gov/i131. Retrieved 2007-11-14.
- ^ "Individual Dose and Risk Calculator for Nevada Test Site fallout". National Cancer Institute. 10/01/07. http://ntsi131.nci.nih.gov/. Retrieved 2007-11-14.
- ^ http://www.thebostonchannel.com/r-video/27338488/detail.html
- ^ "Traces of radioactive iodine found in Washington state milk" Los Angeles Times
- ^ http://www.thyroid-info.com/potassium-iodide.htm
- ^ http://ehp03.niehs.nih.gov/article/fetchArticle.action?articleURI=info:doi/10.1289/ehp.02110927
- ^ "Medical isotopes the likely cause of radiation in Ottawa waste". CBCnews. 04/02/09. http://www.cbc.ca/canada/story/2009/02/04/ot-090204-isotopes.html. Retrieved 2009-02-09.
- ^ "Radioiodine Therapy: Information for Patients". AACE. 2004. http://www.kumc.edu/endocrine/Radioiodine_Therapy.pdf.
- ^ "Instructions for Receiving Radioactive Iodine Therapy after a Thyroid Cancer Survey". University of Washington Medical Center. http://uwmedicine.washington.edu/PatientCare/MedicalSpecialties/SpecialtyCare/UWMEDICALCENTER/Radiology/instructionsthyroidcancersurvey.htm. Retrieved 2009-04-12.
- ^ "Precautions after Out-patient Radioactive Iodine (I-131) Therapy". Department of Nuclear Medicine McMaster University Medical Centre. http://www.hamiltonhealthsciences.ca/documents/Patient%20Education/I131RadioactiveIodineTherapyHHS-trh.pdf.
- ^ Biosafety Manual for Perdue University. Indianapolis. 2002. pp. 7. http://www.ehs.iupui.edu/biohaz_manual/biosafety_manual_v0502.pdf.
- ^ Sutton, Jane (2007-01-29). "Radioactive patients". reuters. http://www.reuters.com/article/health-SP-A/idUSN2633076820070209?pageNumber=2. Retrieved 2009-05-15.
External links
- ANL factsheet
- RadiologyInfo - The radiology information resource for patients: Radioiodine (I -131) Therapy
- Case Studies in Environmental Medicine: Radiation Exposure from Iodine 131
- Sensitivity of Personal Homeland Security Radiation Detectors to Medical Radionuclides and Implications for Counseling of Nuclear Medicine Patients
- NLM Hazardous Substances Databank – Iodine, Radioactive
| Lighter: 130I |
Iodine-131 is an isotope of iodine |
Heavier: 132I |
| Decay product of: 131Te (β-) |
Decay chain of Iodine-131 |
Decays to: 131Xe (β-) |
|
||||||||||||||||||||||||||||||||
|
||||||||||||||
|
||||||||||||||||||||||||||||||